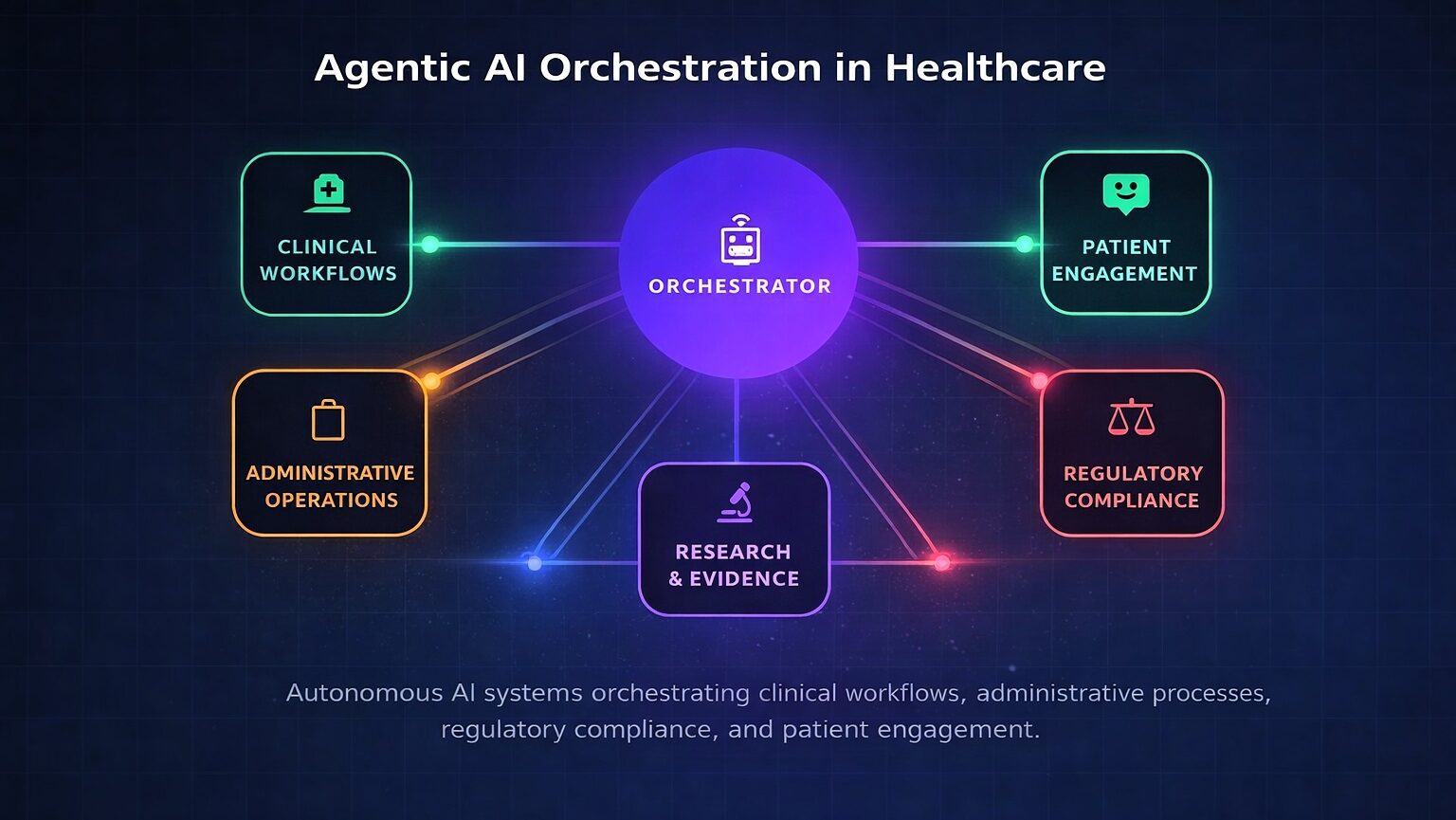
Fig. 1: Agentic AI orchestration across clinical, administrative, regulatory, and patient engagement domains.
The healthcare industry has entered a transformative era. Agentic artificial intelligence — systems capable of autonomous reasoning, planning, and multi-step task execution with defined human oversight — is transitioning from research concept to enterprise deployment.
Unlike traditional machine learning that excels at narrow pattern recognition or generative AI that produces content reactively, agentic AI operates with goal-directed autonomy: decomposing complex objectives, coordinating specialized agents across disparate systems, and adapting strategies based on outcomes. This paradigm shift addresses healthcare’s most persistent challenges, including administrative burden consuming 20 percent of institutional budgets, physician burnout, and the growing complexity of clinical decision-making.1
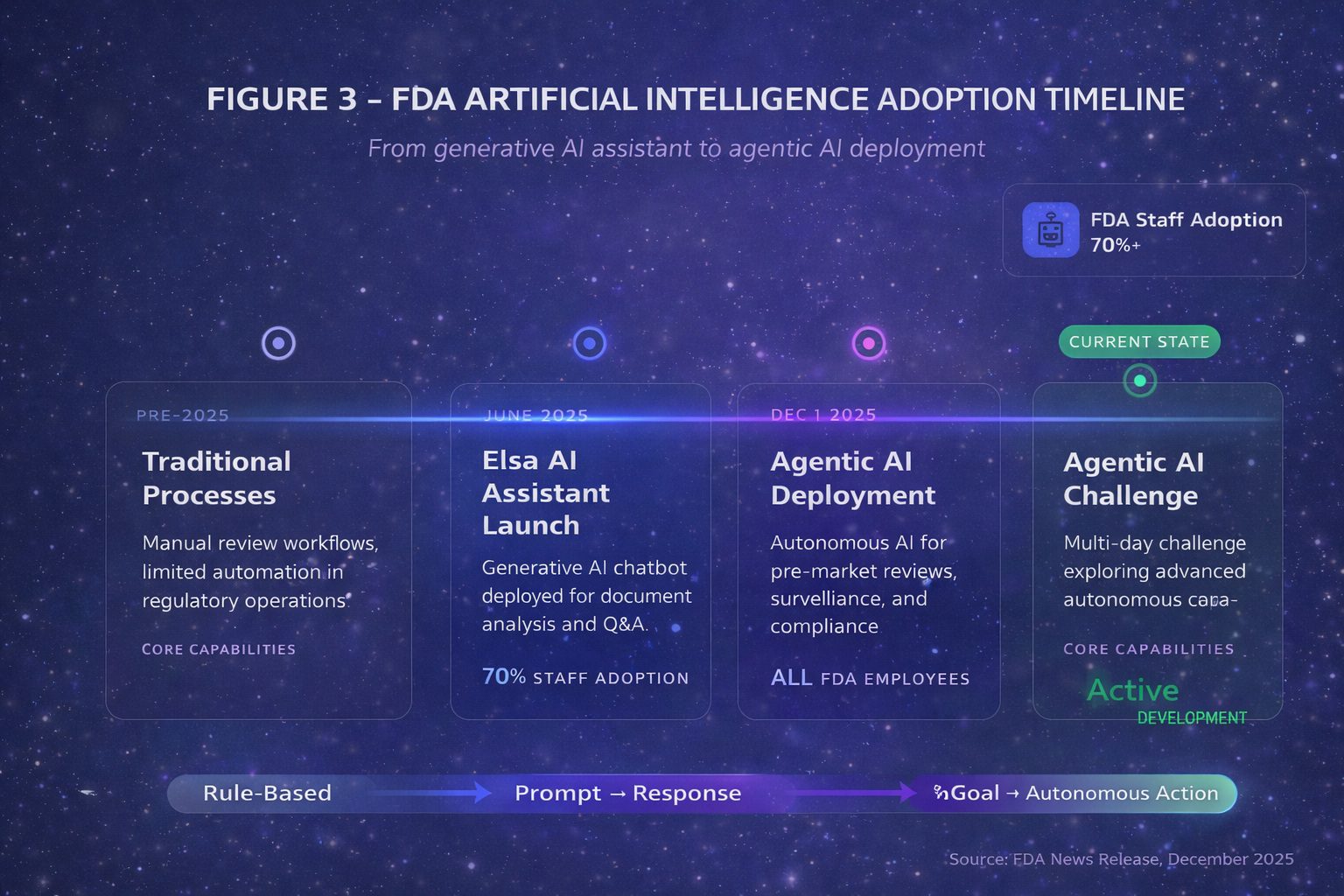
The acceleration in late 2025 has been remarkable. On December 1, 2025, the U.S. Food and Drug Administration announced agentic AI deployment for all agency employees — the first major regulatory body to institutionalize autonomous AI workflows for administrative functions including meeting management, document processing, and compliance operations.2 Critically, these internal tools support agency operations but do not autonomously render pre-market review decisions. Days later, the Department of Health and Human Services released its comprehensive AI strategy positioning autonomous systems as central to federal health operations.3
Defining the Agentic Paradigm
The distinction between agentic AI and its predecessors is substantive. Traditional machine learning excels at classification within narrow domains. Generative AI expanded to content creation. Agentic AI introduces systems that pursue defined goals with limited supervision, typically employing multiple specialized agents coordinated through an orchestration layer.4

Technical precision requires distinguishing workflow automation from true goal-directed autonomy. Research published in Frontiers in Artificial Intelligence found that agentic architectures can reduce cognitive workload by up to 52 percent compared to traditional clinical decision support.1
The Enterprise Technology Landscape
The market is consolidating around major platforms. Microsoft’s healthcare agent orchestrator provides pre-configured agents for clinical trial matching and tumor board preparation. Epic Systems has deployed multiple AI agents: Emmie (patient engagement), Art (provider communications), and Penny (revenue cycle management).6
Table 1: Healthcare Agentic AI Platform Comparison (January 2026)
| Platform | Capabilities | Deployment Considerations |
|---|---|---|
| Microsoft Healthcare Agent Orchestrator | Multi-agent orchestration, clinical trial matching | Azure AI Foundry; broader EHR interoperability |
| Epic (Emmie, Art, Penny, Cosmos) | Patient engagement, provider communications, revenue cycle | Version-dependent; 6-12-month configuration |
| Nuance Dragon Copilot | Ambient clinical and nursing documentation | Generally available; multi-EHR integration |
| Atropos Evidence Agent | Proactive real-world evidence at point of care | Pilot deployments; addresses evidence accessibility |
Regulatory Landscape and Liability
The FDA’s database lists over 1,250 AI-enabled medical devices authorized for marketing, but the vast majority are narrow diagnostic imaging tools. True agentic systems for clinical decision-making face the most stringent Class III regulatory pathway.9
The liability landscape presents novel challenges. Traditionally, the “learned intermediary” doctrine shields device manufacturers when physicians exercise independent judgment. However, agentic systems that execute autonomously may eliminate this shield, exposing manufacturers to direct liability.12
Implementation Prioritization Framework
Table 2: Implementation Prioritization Framework
| Phase | Use Cases | Risk Profile | Timeline |
|---|---|---|---|
| Immediate (2026) | Scheduling, prior auth, messaging triage | Low clinical risk | 3-6 months |
| Near-term (2026-2027) | Ambient documentation, coding assistance | Moderate risk | 6-12 months |
| Medium-term (2027-2028) | Clinical decision support, care coordination | Higher risk | 12-24 months |
| Longer-term (2028+) | Autonomous monitoring, closed-loop systems | Highest risk | 24+ months |
References
- Hinostroza Fuentes N, et al. Frontiers in Artificial Intelligence. 2025;8.
- U.S. Food and Drug Administration. News Release. Dec 1, 2025.
- HHS Artificial Intelligence Strategy. Dec 4, 2025.
- IBM. What is Agentic AI? 2025.
- Microsoft Build 2025 Announcement.
- Healthcare IT News. Epic UGM 2025.
- Microsoft Industry Blog. Nov 18, 2025.
- PDA News Brief. Dec 2025.
- Bipartisan Policy Center. Nov 10, 2025.
- European Pharmaceutical Review. Jan 2026.
- Manatt Health AI Policy Tracker. 2025.
- Price WN, et al. JAMA. 2019;322(18):1765-1766.
- Parasuraman R, Human Factors. 2010.
- Longoni C, Journal of Consumer Research. 2019.
- Medical Economics. Johns Hopkins study. 2025.
- Health Affairs. 2014;33(9):1586-1594.
- Gartner Top Strategic Trends 2025.
- Microsoft Research Podcast. July 23, 2025.

About the Author
Dr. Srikanth Mahankali is a leading expert in the implementation of medical AI and policy. As CEO of Shree Advisory & Consulting and a member of the NSF/MITRE AI Workforce Working Group, he shapes national AI strategy while driving responsible innovation.